By Samanta Adam
Introduction
The control of degradation products in pharmaceuticals remains a challenging task among pharmaceutical scientists.
The forced degradation guideline recommended by the International Conference on Harmonization (ICH) is used to identify potential degradation products, which are formed during stress degradation, but are actually formed in a drug substance or in the Product, due to storage conditions during its shelf life. This information helps scientists to design appropriate measures to minimize expected impurities in the life cycle of drug products.
Stress testing (forced degradation study) has become an important regulatory requirement fundamental to the drug development and approval process. It provides evidence on the impact of various environmental factors on the quality of the drug, drug substance or drug product in real time and is the scientific basis for recommending storage conditions and package technology for a dosage form. In pharmaceutical analytical research, the design of stress conditions is crucial and the preliminary step in conducting stability studies for the drug or product, because it reveals the intrinsic mechanism of drug and decomposition along with information regarding the percentage of degradation and number of impurities expected in the life cycle of the drug.
In the literature (Brattty M., 2020) we can also find that forced degradation studies are performed using a chemical reagent or stress inducer in each type of stress condition including: acid and basic hydrolysis, oxidation and photolysis.
Oxidation reactions
Free radicals such as those generated from hydrogen peroxide and singlet oxygen can oxidize drugs. Oxidation involves the loss of electrons and can degrade the drug structure.
Free radicals are normally formed as a by-product of oxygen metabolism in animals and plants. Drugs, when metabolized, can produce free radicals. Light can oxidize drugs and accelerate their degradation (Kryczyk-Poprawa A., 2020).
One of the typical reactions that can occur in drug structures are as follows:
-Epoxy Formation
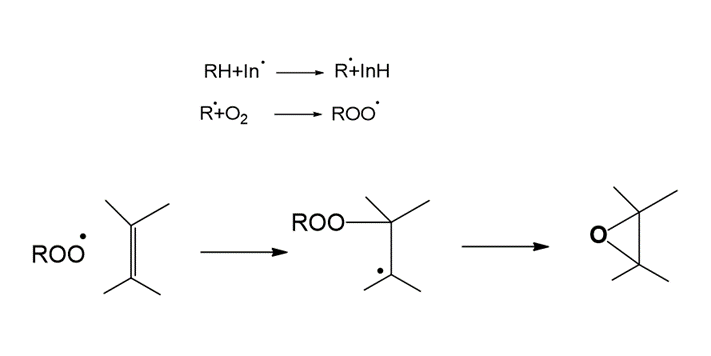
Figure 1. General reaction mechanism of Epoxide formation.
For the formation of the epoxide, the free radical attacks the double bond in the molecule, generating a tertiary radical, which gives way to the formation of the epoxide.
For example, the formation of the Epoxide in the Tetrazepam molecule.
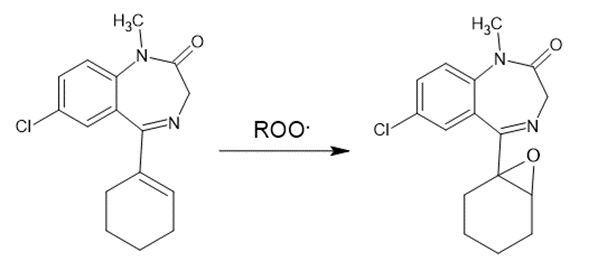
Figure 2. Oxidation of Tetrazepam.
-Formation of N-Oxides
Tertiary amines are oxidized in the presence of oxidizing agents to form N-oxides.
For example, the formation of N-oxide in the oxidation of the Pantoprazole molecule (Bratty M., 2020).
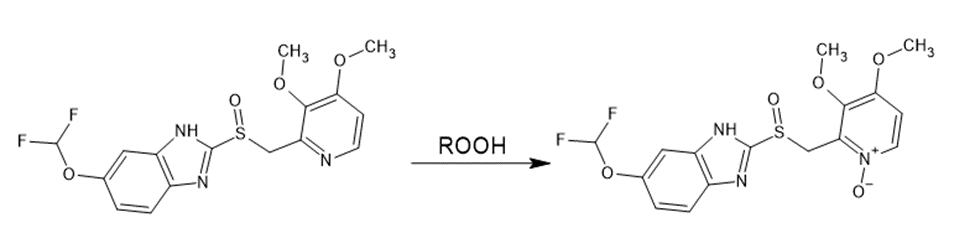
Figure 3. Oxidation of Pantoprazole.
-Oxidation of alcohols.
Alkanes can be oxidized to alcohols and these in turn can be oxidized to aldehydes or ketones. Oxidation occurs in conjunction with the loss of a hydrogen molecule. This is why those carbons that have no hydrogens cannot be oxidized, since they have no hydrogen bonds to lose.
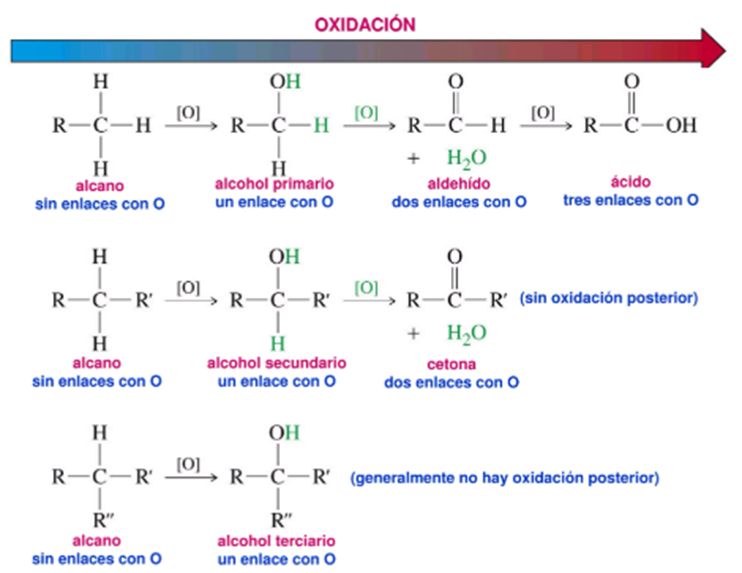
Figure 4. Oxidation of alcohols.
For example, the oxidation of the Dexamethasone molecule, where in a first step the primary alcohol is oxidized to release aCO2 molecule, subsequently the resulting aldehyde is oxidized to release the secondCO2 molecule and finally form the corresponding ketone (Ummiti K., 2014).
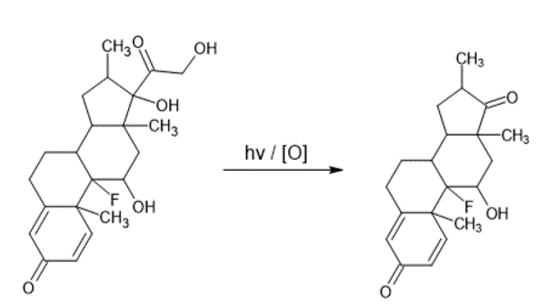
Figure 5. Oxidation of Dexamethasone.
How to prevent drug degradation?
-Hermetic containers:
Storing drugs in airtight containers can prevent exposure to light and air, thus preventing drug oxidation and the formation of free radicals.
-Dry environment:
Storing drugs in a dry environment can prevent hydrolysis and oxidation of the drugs.
-Antioxidants:
The addition of antioxidants in the drug formulation can prevent oxidation of the drug, e.g., Sodium Sulfite, Sodium Metabisulfite, Sodium Thiosulfate, Ascorbic Acid, Ascorbyl Palmitate (AP), Propyl gallate (PG), Butylated hydroxytoluene (BHT), Butylated hydroxyanisole (BHA), Alpha-tocopherol (α-TOH), Tartaric Acid, Citric Acid, Ethylenediaminetetraacetic Acid (EDTA), etc (Zuñiga S., 2021).
Bibliography
- FDA Guidance for Industry (2003), INDs for Phase II and III Studies-Chemistry, Manufacturing, and Controls Information, Food and Drug Administration. https://www.fda.gov/drugs.
- International Conference on Harmonization, ICH Q2 Guidelines (2003), Available from: http://www.fda.gov/downloads/RegulatoryInformation/Guidances/ucm128204.pdf.
- Bratty M., Thangavel N., Peraman R., Kumar V. (2019). HPLC-DAD Method for Investigating Pantoprazole for its Stress-Dependent Degradation by Photolysis and Oxidation. Acta Chromatographic 32(2020)4, 247-255.
- Kryczyk-Poprawa A., Kwiecién A., Opoka W., (2019). Photostability of Topical Agents Applied to the Skin:A Review. Pharmaceutics 2020, 12, 10; doi:10.3390/pharmaceutics12010010.
- Ummiti K., Vakkala S., Panuganti V., Reddy M. (2014). ISOLATION, IDENTIFICATION, AND
CHARACTERIZATION OF 17-OXODEXAMETHASONE, AN OXIDATIVE DEGRADATION IMPURITY OF DEXAMETHASONE USING FLASH CHROMATOGRAPHY AND NMR/HRMS/IR. Journal of Liquid Chromatography & Related Technologies, 37:17, 2403-2419.
- Zuñiga S, Paulina S., (2021). Review of the most commonly used antioxidants in pharmaceutical formulations available in the market. https://repositorio.xoc.uam.mx/jspui/handle/123456789/38105
