By Arturo Rodríguez
Dexamethasone is a synthetic glucocorticoid, i.e., it is similar to a natural hormone produced by the adrenal glands, used primarily for its potent anti-inflammatory effect, so it is used to treat certain forms of arthritis; skin, blood, kidney, eye, thyroid, and intestinal disorders (e.g., colitis); severe allergies; and asthma. Dexamethasone is also used to treat certain types of cancer (MedlinePlus, 2023).
Although its anti-inflammatory activity is considerable even at low doses, it maintains little effect on electrolyte balance. At doses of equal anti-inflammatory potency, dexamethasone lacks almost completely the sodium-retaining property of hydrocortisone and closely related derivatives.
Glucocorticoids have intense and varied metabolic effects and, in addition, modify the body's immune response to various stimuli.
Dexamethasone has a ketone group that is susceptible to Baeyer-Villiger type oxidation reactions in the presence of oxidizing agents and Lewis acids to generate an ester that subsequently undergoes hydrolysis that can be carried out in a basic or acid medium, catalyzed by temperature to generate a carboxylic acid, so the reaction is carried out in two stages (ten Brink, Arends, & Sheldon, 2004).
In the first stage the electronic pair of the oxygen of the carbonyl group of Dexamethasone performs a nucleophilic attack on the Lewis acid, which are species capable of accepting an electronic pair such as protons, heavy metals, transition metals, among others; This attack generates an electronic deficiency in the oxygen and subsequently the delocalization of the carbon-carbonyl-oxygen double bond towards the latter to stabilize the charge, thus generating a secondary carbocation which undergoes a nucleophilic attack by the oxidant (e.g. hydrogen peroxide), when the oxidizing oxygen-carbocation bonding is carried out, an electronic deficiency is produced in the bonding oxygen, so in order to stabilize the charge, a heterolytic rupture is favored between the bonding oxygen and the hydrogen that is bonded to it to stabilize the charge, at the same time that by means of a migration the R group bonded to carbonyl is joined to the oxygen of the oxidizing agent, which produces a primary carbocation, The Lewis acid then undergoes a heterolytic cleavage to generate an electronic excess on the carbonyl oxygen which immediately delocalizes to re-form the double bond and at the same time the hydroxyl group of the oxidant leaves as a good leaving group to generate an ester.
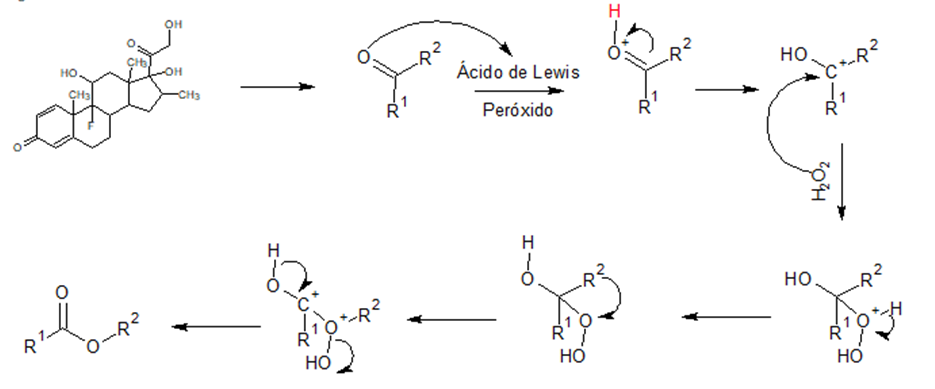
Subsequently, during the second stage, a nucleophilic attack is performed by the presence of a base (such as sodium hydroxide) if it is a basic hydrolysis to the carbonyl carbon, producing a delocalization of the double bond towards the carbonyl oxygen, by the action of temperature there is a heterolytic rupture between the oxygen of the ester and the carbonyl carbon producing a basic hydrolysis and a primary carbocation that is immediately stabilized by the delocalization of the electronic pair supported by the oxygen which returns to form the double bond of the carbonyl group. This generates the carboxylic acid and methoxide which enter into an acid-base equilibrium by exchanging the proton of the carboxylic acid (Fernandez, 2023).
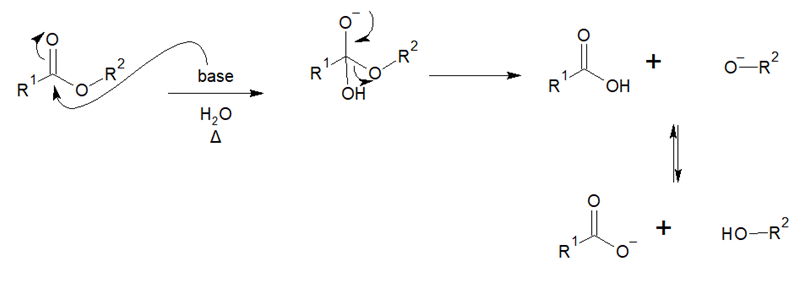
In Dexamethasone formulations, acid Dexamethasone may present as a degradation product generated by API-excipient interactions that may be counterproductive for the manufacturer and result in reformulation processes if the limits outlined in ICH Q3A and ICH Q3B Guidelines are exceeded.
At QSAR Analytics we help pharmaceutical companies to carry out the process, identification and qualification of impurities and degradation products, our work is in full compliance with the ICH Q3A and ICH Q3B Guidelines which harmonize the acceptable levels of organic impurities from synthesis and degradation.
Bibliography
- United States Pharmacopoeia (2023). Dexamethasone Sodium Phosphate. doi:https://doi.usp.org/USPNF/USPNF_M23450_05_02.html
- European Pharmacopoeia (2020). Information leaflet Ph. Eur. Reference Standard: Dexamethasone soduim phosphate for peak identification CRS batch 2. EDQM. Retrieved 2023, from https://crs.edqm.eu/db/4DCGI/View=Y0001477
- Fernández, G. (2023). Basic hydrolysis of esters. Saponification. Retrieved 2023, from https://www.quimicaorganica.org/esteres/449-hidrolisis-basica-de-esteres-saponificacion.html
- MedlinePlus (2023). Dexamethasone. Retrieved 2023, from https://medlineplus.gov/spanish/druginfo/meds/a682792-es.html
- ten Brink, G. J., Arends, I., & Sheldon, R. A. (2004). The Baeyer-Villiger reaction: new developments toward Greener procedures. Chemical Reviewa(9), 4105-4123. doi:https://doi.org/10.1021/cr030011l
