By Daniela Alva
An elimination reaction is one in which one or two substituents are removed from the original molecule. The elimination can proceed through a one- or two-step mechanism, known as E1 and E2 elimination, respectively. In both types of mechanisms the elimination results in the formation of a double bond, a triple bond or a ring (Li, 2012). In the E1 elimination mechanism, an acidic medium and a leaving group on the substrate molecule are required for the reaction to proceed, whereas in the E2 elimination mechanism, a leaving group and a nucleophile, as well as a basic pH are required for the reaction to complete.
The most relevant drug degradation that takes place through an elimination mechanism is dehydration or water loss. Several corticosteroids such as dexamethasone, mometasone, betamethasone and prednisone undergo a dehydration reaction, particularly under acidic conditions (Li, 2012). In a pharmaceutical form, some excipients may have traces of acids that favor the reaction, for example, in lactose monohydrate an autooxidation reaction may occur, causing traces of formic acid to form, which confer acidity to the medium and allow this type of mechanism to take place.
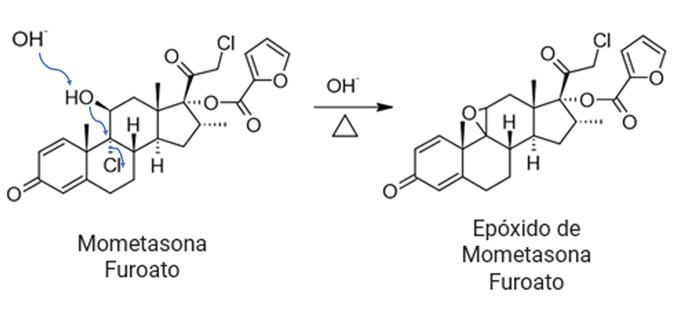
Another reaction involved in elimination is dehydrohalogenation. Mometasone furoate, a corticosteroid prodrug widely used for a variety of anti-inflammatory indications, has a chlorine atom in its molecular structure, which is susceptible to dehydrochlorination (Li, 2012), since the halogen being an electronegative atom is a good leaving group, it also has alcohol groups that can be eliminated by a dehydrohalogenation reaction. One of the products resulting from these eliminations is mometasone fluorate epoxide (Li, 2012), when the molecule is in basic media an E2 elimination mechanism can occur resulting in the formation of a 3-membered ring (epoxide), since chlorine and water can be easily removed from the structure. Some excipients such as sodium hydroxide can catalyze an E2 elimination mechanism, as the hydroxyl groups can act as nucleophiles. Similarly, excipients containing hydroxyl groups such as ethanol, glycerol and methanol can act as nucleophiles. In the case of E1 elimination, some excipients can favor the reaction by having traces of acids involved in the mechanism, such is the case of magnesium stearate that can present traces of formic acid resulting from its autooxidation.
Bibliography:
- Li, M. (2012). Organic Chemistry of Drug Degradation. RSC Publising. United Kingdom.
