At QSAR Analytics we help pharmaceutical companies with nitrosamine formation risk analysis, especially when they face constraints such as lack of time, expert staff or urgency. We enable our clients to meet regulatory agency requirements and keep their drugs on the market.
What are Nitrosamines?
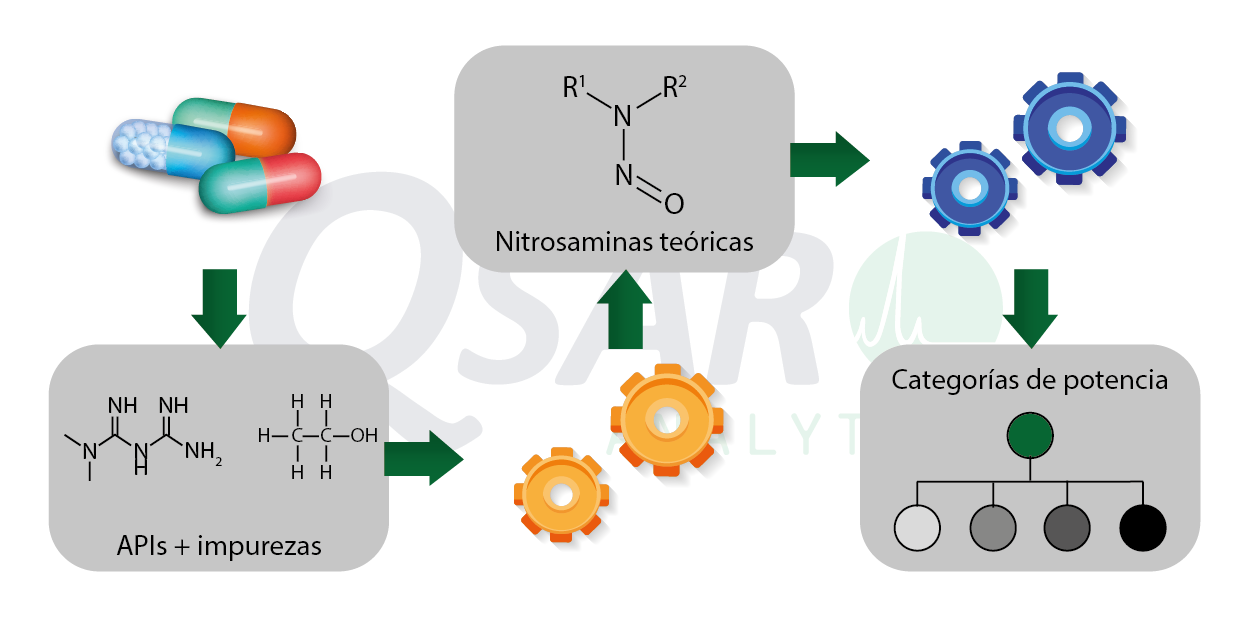
Nitrosamines are potentially carcinogenic compounds that can be formed during API synthesis, storage or by generation in the drug manufacturing process by various means. Today this family is being studied throughout the world to ensure that patients have safe medicines.

Our Approach
Based on COFEPRIS guidelines, our risk analysis (step 1 of the guide) follows a detailed process to ensure product quality:
- Risk Identification: We detect possible sources of nitrosamine formation, such as reagents and excipients used in API synthesis or manufacturing processes.
- Risk Assessment: We quantify the level of risk associated with the formation of nitrosamines using advanced tools such as QSAR models and in silico analysis to predict the formation of these impurities.
- Risk Focus: We point out the critical areas where attention must be paid to reduce nitrosamine levels, helping the client to make informed decisions on the actions to be implemented.
- Toxicological data and PT limits. We provide toxicological information, either by databases or in silico studies, to generate maximum impurity percentages for each drug evaluated.
Benefits of choosing QSAR Analytics as your partner for this analysis
- Specialization in Impurities: We are experts in impurities and national referents in organic impurities studies, we have been working since 2021 in the identification and evaluation of nitrosamines for clients in other countries.
- Total Compliance: Our knowledge, strategic allies and experience guarantee compliance with COFEPRIS regulations.
- Multidisciplinary Team: We are specialized minds focused on helping to solve a single problem, "nitrosamines".
- Technology: We use (Q)SAR models and specialized databases to support the study.
- Efficient response times: We know that time is a critical factor to comply with the requirements of the authority, this is clear to us and therefore, we have the mission to share this responsibility with each of our clients.
