By Nayeli Ramirez
Most drug degradation mechanisms include hydrolysis/dehydration, oxidation, isomerization/epimerization, decarboxylation, rearrangement, dimerization/polymerization and photolysis reactions. Some reactions, also, involve interaction with some excipients.
The formation of degradation products can take place under different conditions, including acidic, basic, oxidative and photolytic conditions. Hydrolysis reactions can be catalyzed by the effect of temperature, while oxidation reactions are carried out through the presence of radicals, as are photolytic reactions.
Amines are a very common functional group in drugs and are susceptible to different degradation reactions. Amines can be primary, secondary and tertiary and when deprotonated they can act as nucleophiles, aldehydes being a functional group with which they can easily react.
The Maillard reaction is a set of complex reactions that takes place between amines and reducing sugars in their aldol form, resulting in the formation of brown stains or pigmentation in drugs.
The first step corresponds to the interaction between a reducing sugar (e.g. lactose) and a primary amine, which will result in the formation of an iminium ion. The iminium ions are unstable and can tautomerize to form an α-amino ketone, the product formed is known as "Amadori rearrangement" (ARP). The following figure shows the general mechanism of how the reaction is carried out (Baertschi, 2005).
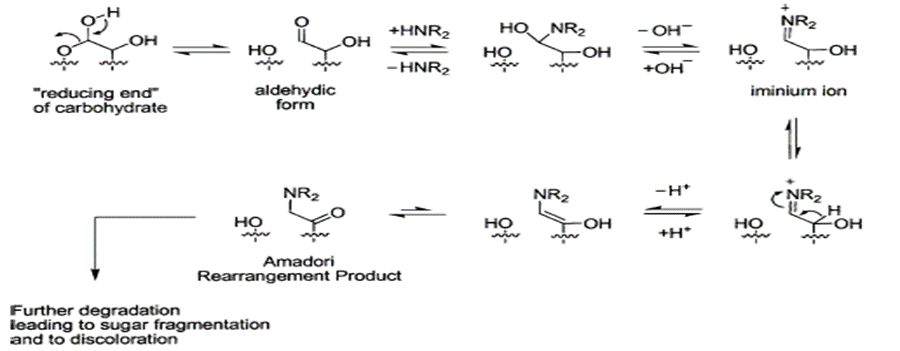
General scheme of the Maillard reaction, obtained from Baerthshi, 2005).
PRA is an important intermediate as an initial stage, since in some storage conditions it can be generated and accumulate as an impurity, and can be observed as different peaks in chromatograms. In an intermediate stage, dehydration reactions and fragmentation of sugars can take place, which will give as final product melanoidins, which can be perceived as yellow and brown pigmentations in drugs.

Figure 2. Tablets with characteristic Maillard reaction marks.
The Maillard reaction is more likely to take place when sugars are in their amorphous form compared to their crystalline form. Since it is a pH-dependent reaction, it is recommended to maintain a neutral pH in the environment (Baertschi, 2005).
Bibliography
- Baertschi, S. (2005). Pharmaceutical Stress Testing. USA. Taylor and Francis.
