By José Juan Martínez
As we know, drug stability is the ability of the drug within a sealed container to maintain over a period of storage and use the established quality specifications.
During this stability period, degradation products may be formed due to chemical changes in the drug, influenced by certain factors such as light, temperature, pH, water, by reaction with an excipient or with the container-seal system (Secretaría de Salud [SSA], 2015).
Among the most common reactions in this type of studies are decarboxylation reactions, although not limited to be observed exclusively in degradations, the reaction is used in a wide variety of drugs for the conversion of inactive precursors to the active form that is responsible for the therapeutic effect, improvement of bioavailability by converting the active into a more soluble form, as well as modifying the duration of action of a drug while it is metabolized in the body.
"Susceptibility to decarboxylation depends on the type of compound and its chemical structure. The β-ketoesters can undergo decarboxylation and yield a ketone." (Stanley H. Pine, Jemes B., Donald J. Cram & George S. Hammond, 1988). To understand this process and how it could affect the quality and stability of drugs, generate unwanted degradation products and ensure that the use of drugs is safe, we must first understand What is decarboxylation, where in a general way the loss of a carbon dioxide molecule in a carboxylic acid is called decarboxylation.
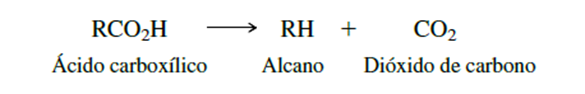
Figure 1. Decarboxylation reaction (Carey, 2006).
"Decarboxylation is a chemical reaction that allows to structurally transform a molecule, which involves the removal of a carboxyl group (-COOH) from a molecule, with the particular breaking of the bond between a carbon atom and a carboxyl group, loss of carbon dioxide (-CO2) and the formation of a new substance such as a ketone. This reaction can be generated by applying appropriate temperature which varies according to the R group attached to the carboxyl, catalysts or enzymatic reactions, but depends on the molecule and the specific process. As an example, if there were a carbonyl group in the β-position relative to the carboxyl group, decarboxylation occurs easily, simply by gentle heating" as in Figures 2 and 3 (Stanley H. Pine, Jemes B., Donald J. Cram & George S. Hammond, 1988).
"Decarboxylation of simple carboxylic acids takes place with great difficulty and is rarely observed" whereas compounds that undergo thermal decarboxylation readily comprise those related to malonic acid."
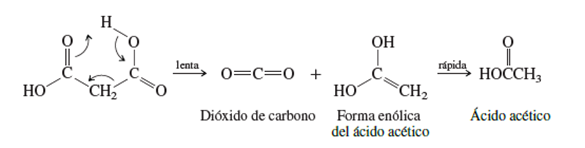
Figure 2. Decarboxylation of malonic acid and related compounds (Carey, 2006).
"The transition state involves the carbonyl oxygen of a carboxyl group, which remains behind, acting as a proton acceptor for the hydroxyl group of the carboxyl that is lost. Cleavage of the carbon-carbon bond leads to the enolic form of acetic acid, along with a carbon dioxide molecule. The enolic intermediate then tautomerizes and forms acetic acid" (Carey, 2006).
It is important to keep in mind that not all molecules that feature carboxylic acids can undergo decarboxylation, so "decarboxylation is not a general reaction of carboxylic acids, rather, it is unique to compounds that have a second carbonyl group two atoms away fromCO2H". That is, only substituted malonic acids and β-keto acids experience loss ofCO2 upon heating. The reaction occurs by a cyclic mechanism and involves the initial formation of an enol, thus explaining the need to have a second carbonyl group in the appropriate position (McMurry Jhon, 2008).
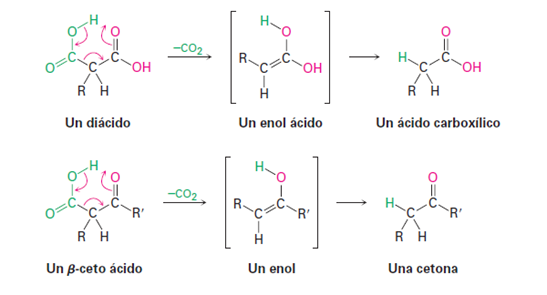
Figure 3. Decarboxylation reaction (McMurry Jhon, 2008).
The decarboxylation reaction is also a critical process in the synthesis route for the production of active ingredients used for the manufacture of drugs, due to its impact on the efficacy, safety and quality of the products obtained, and can favor the creation of more efficient products with the desired therapeutic effect. However, it should be borne in mind that in the presence of errors during the process, potentially dangerous drugs may result.
Decarboxylation reactions can influence drug stability. Leading to drug degradation, affecting efficacy and shelf life. Therefore, it is important to understand and control this process during drug development and storage. This can be achieved by choosing suitable precursors, adjusting reaction conditions and protecting functional groups when necessary.
In summary, it can be considered that decarboxylation reactions are favored by the use of temperature and that a carbonyl in the β-carboxylic position is necessary, so that only substituted malonic acids and β-ketoacids experienceCO2 loss upon heating.
Bibliography
- Carey, F. A. (2006). Organic Chemistry (6th ed.). Mexico D.F.: Mc Graw Hill.
- McMurry Jhon (2008). Organic Chemistry (7th ed.). Mexico, D.F.: Cengage Learning.
- Ministry of Health [SSA] (2015). NORMA Oficial Mexicana NOM-073-SSA1-2015, Stability of drugs and medicines, as well as herbal remedies. Retrieved on 09/26/2023, from https://dof.gob.mx/nota_detalle.php?codigo=5440183&fecha=07/06/2016#gsc.tab=0
- Stanley H. Pine, Jemes B., Donald J. Cram & George S. Hammond (1988). Organic Chemistry (4th ed.). Mexico: Mc Graw Hill.
