An analytical method is a sequence of steps that allows us to have our sample in the necessary conditions to obtain information about its composition, structure, and/or properties; this method can be classified according to the type of analysis performed, as qualitative or quantitative; also by the type of analytical technique used to determine the characteristics of the sample, which can be spectrophotometric, chromatographic, microbiological, electrochemical, among others.
One of the most important challenges in developing an analytical method to quantify impurities with HPLC is to have molecules that have few chromophore groups. Chromophore groups are those that absorb electromagnetic radiation in the visible or ultraviolet range. Radiation absorption is the physical principle that allows detection and measurement of the separated components by HPLC using a UV-VIS detector. When a molecule has few chromophore groups, it means that it has a low radiation absorption, which makes its detection and quantification difficult.
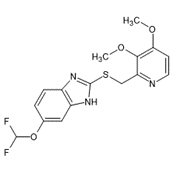
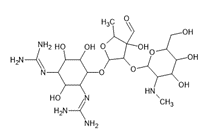
Fig. 1 Molecule with several chromophore groups Vs Molecule with few chromophore groups.
What do I do when I have a molecule that has few chromophore groups?
To overcome the challenge of having molecules that have few chromophore groups, several strategies have been proposed that when added together can support you in the development of an analytical method. We will first look at the strategies that you can do with the design of the analytical technique and then those that can modify your analyte.
Analytical technique design
Reagents: Verify that the cut-off length of your reagents does not interfere with that of your analyte, remember that this is the wavelength at which the solvent absorbs a significant portion of the light, causing a loss of signal and inability to run an analysis. At this point you will have to evaluate whether it is in your best interest to lose absorptivity, but avoid interference, or if the opposite is the case. If the latter is the case, consider purchasing MASS grade reagents.
pH of the mobile phase: At this point it is important to ensure that at the working pH you have 100% of your analyte, in the case of impurity methods in which you do not know the structure, it is important to perform pH tests according to the pKa of your parent analyte. This will not contribute to having more chromophore groups, but it will help you to see your analyte at 100% and not at 75%.
Concentrate your sample:
First option: Know the solubility of your molecule of interest and your excipients, since you must make a mixture in which you can solubilize your analyte, but avoid solubilizing any excipient that might cause interference.
Second option: Increase the injection volume, in this case you must take into account two aspects: the first one is the cleanliness of the injector, so it is important to avoid the use of pre-arranged septum and consider having washing vials. The second aspect is related to the saturation of your column, so it is advisable to use a precolumn and to perform consecutive washes between runs.
Third option: Solid phase extraction (SPE) is a sample preparation technique, it allows extraction, clean-up and concentration of analytes prior to quantification.
Sample cleaning: Centrifuge your sample and test different types of syringe filters, as some may not be effective.
Stationary phase: The selection of the correct stationary phase will prevent the peaks obtained from being tailed, wide or having a fronting. Therefore, it will be indispensable that within your parameters established in suitability you include column efficiency.
Equipment parameters:
It is essential that, before making these changes, you consult with your service technician, as the names may vary depending on the make and model of HPLC equipment, and he will provide you with information on the appropriate parameters and their use.
Auto zero: It will take as target the spectrum of the eluent.
Sampling rate: Too few sampling points result in inconsistencies in areas and heights.
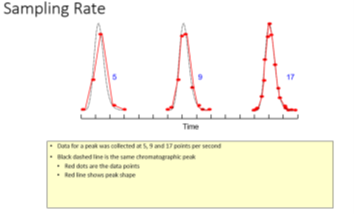
Fig. 2 Sampling rate5
Time constant: Helps to achieve good signal-to-noise by filtering out high frequency noise, however, if this time constant is too high, it will result in distorted peaks.
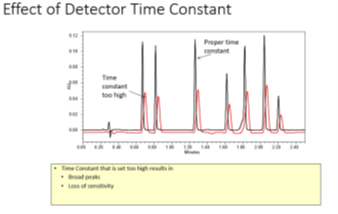
Fig. 3 Time constant5
Noise + Solvent: It is not advisable to move this parameter; however, if you have a very noisy baseline and low absorptivity of the molecule of interest, you may obtain peaks that do not comply with purity, so you should do the steps marked by the service provider so that this calculation with this movement is correct.
Analyte modification
Strategies that modify the molecule consist of altering the chemical or physical structure of the molecule to increase its radiation absorption.
Derivatization: It consists of joining chromophore groups in the molecule of interest by means of a chemical reaction prior or subsequent to HPLC separation.
In this parameter you must be careful with the stability of your sample. Make sure that the chromatographic equipment can perform the function of sample taking and derivatization and mixing if it is a previous chemical reaction. If the reaction is subsequent, you must verify that you have the necessary equipment and space before reaching the detector.
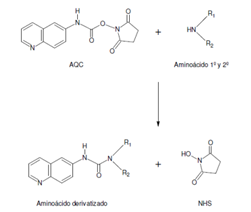
Fig. 4 Derivatization3
Complexation: It consists of forming a complex with another substance that has a higher radiation absorption.
Ionization: which consists of changing the charge state of the molecule by a pH change or an ionic additive. For example, an amine can be ionized with hydrochloric acid to form an ammonium cation that has higher UV absorption.
What do I do when I have implemented all of the above?
You will hardly fall at this point, but if the above modifications did not give you the results you expected, you can consider using a mass method. However, you have to consider that when dealing with a method of unknown impurities, you will have to elucidate their molecular structure. For this, it is advisable to start from the base structure of your main analyte. Capillary voltage changes should be adequate, since a very high one with labile molecules could cause it to fragment from the beginning.
Bibliography
- Avila-Camacho, F. J., Moreno-Villalba, L. M., Cerón-Montes, G. I., Garrido-Hernández, A., Cardoso-Chávez, C. A., & Pacheco-Piña, C. J. (2021). Design of a low-cost UV-VIS spectrophotometer for the biochemical industry: A Review. Pädi Boletín Científico De Ciencias Básicas E Ingenierías Del ICBI, 9(Especial2), 19-28. https://doi. org/10.29057/icbi.v9iEspecial2.778
- Fábio R. P. RochaI,*; Leonardo S. G. TeixeiraII, Estratégias para aumento de sensibilidade em espectrofotometria UV-VIS IInstituto de Química, Universidade de São Paulo, CP 26077, 05513-970 São Paulo - SP IIDepartamento de Engenharia e Arquitetura, Universidade Salvador, Av. Cardeal da Silva, 132, 40220-141 Salvador - BA, Divulgação - Quím. Nova 27 (5) - Out 2004 - https://doi.org/10.1590/S0100-40422004000500021
- Amézquita L. Fernando, Mendoza O. Diana, Basic Course on Ultraviolet-Visible Spectroscopy. University of Guanajuato, Faculty of Chemistry.
- How do you set up the Purity Thresholds in Empower 3? - WKB11215, Waters Knowledge Base
- Setting the Sampling Rate and Time Constant in Empower - Tip207, Waters Knowledge Base.
